Triphenylphosphine as Efficient Antidote for the Sulfur‐Poisoning of the Pd/C Hydrogenation Catalyst - Xiong - 2021 - ChemCatChem - Wiley Online Library
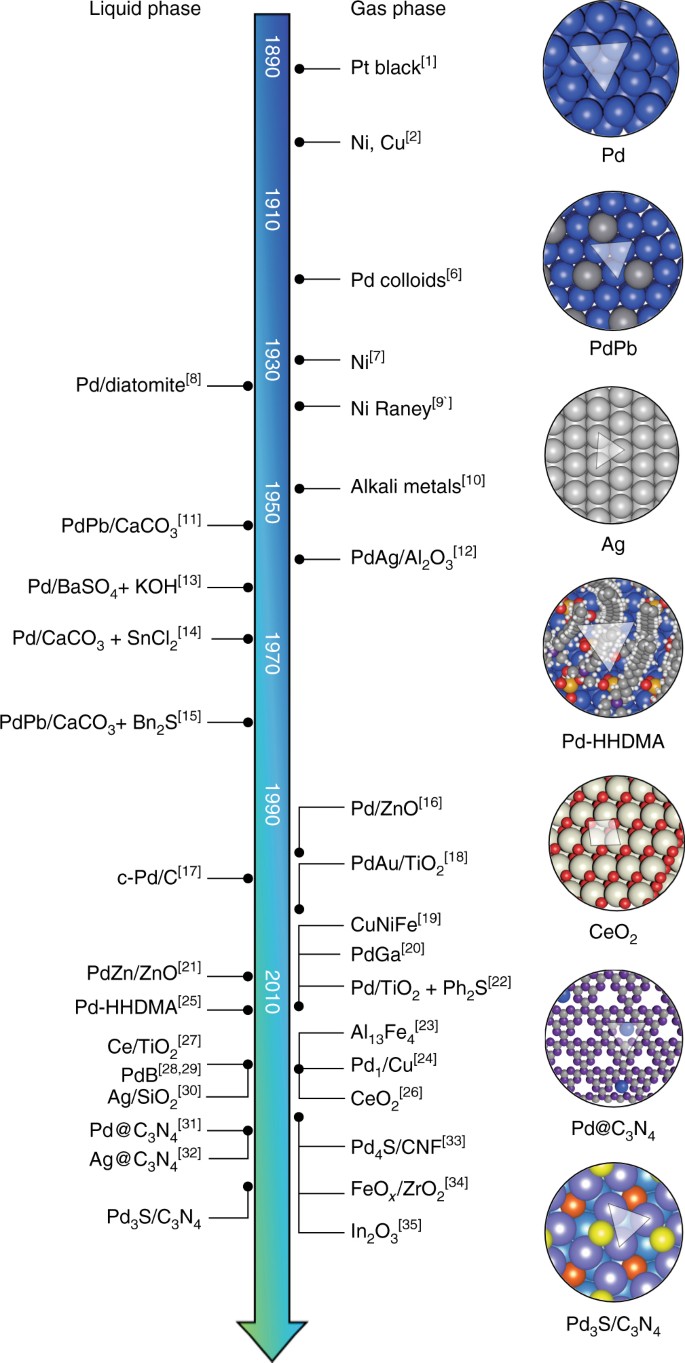
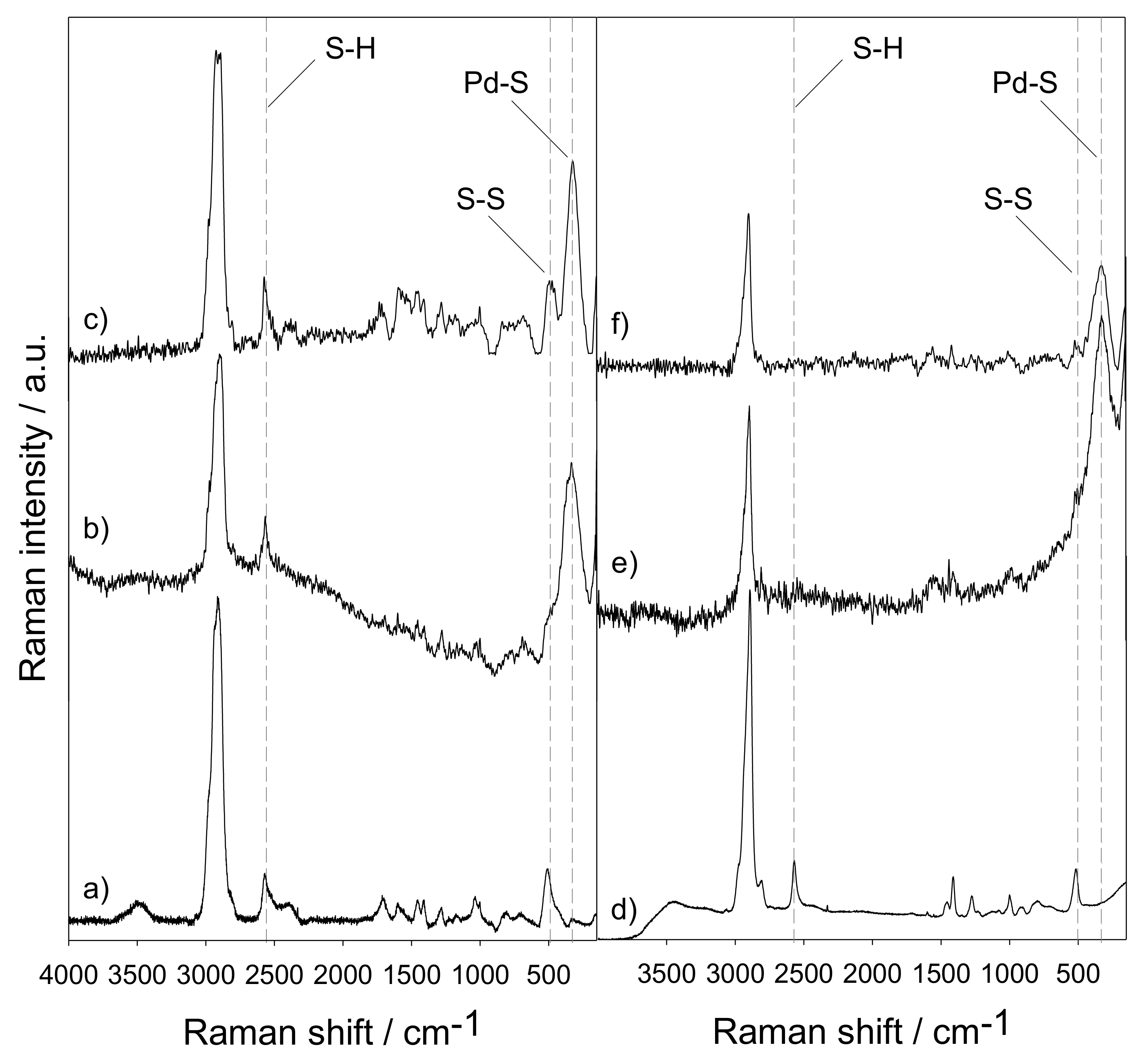
Selective ensembles in supported palladium sulfide nanoparticles for alkyne semi-hydrogenation | Nature Communications

Palladium‐Catalyzed C–S Bond Formation of Stable Enamines with Arene/Alkanethiols: Highly Regioselective Synthesis of β‐Amino Sulfides,European Journal of Organic Chemistry - X-MOL
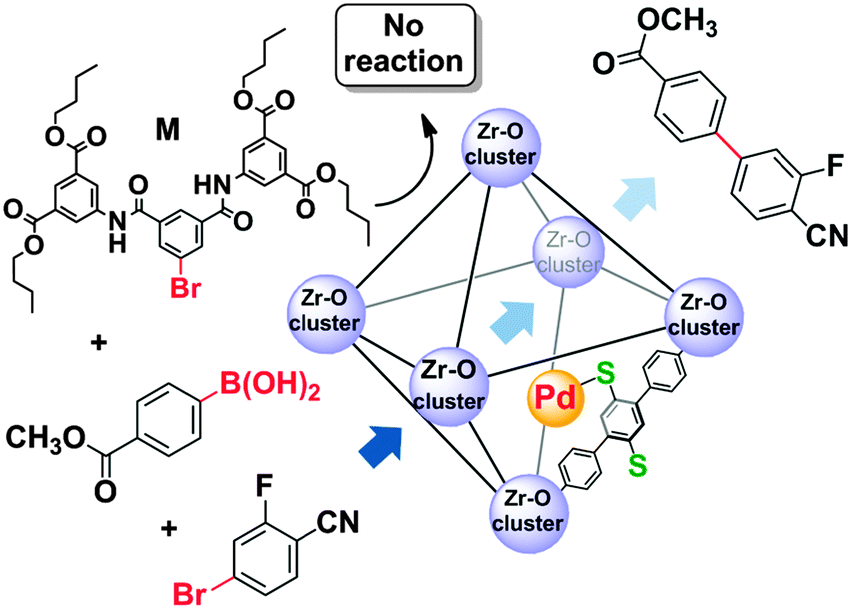
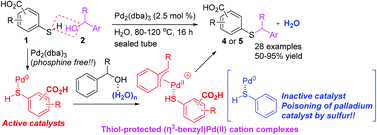
Tackling poison and leach: catalysis by dangling thiol–palladium functions within a porous metal–organic solid - Chemical Communications (RSC Publishing)
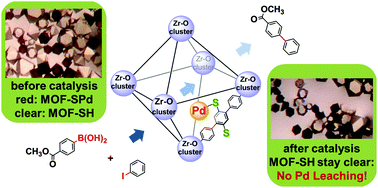
Tackling poison and leach: catalysis by dangling thiol–palladium functions within a porous metal–organic solid - Chemical Communications (RSC Publishing)

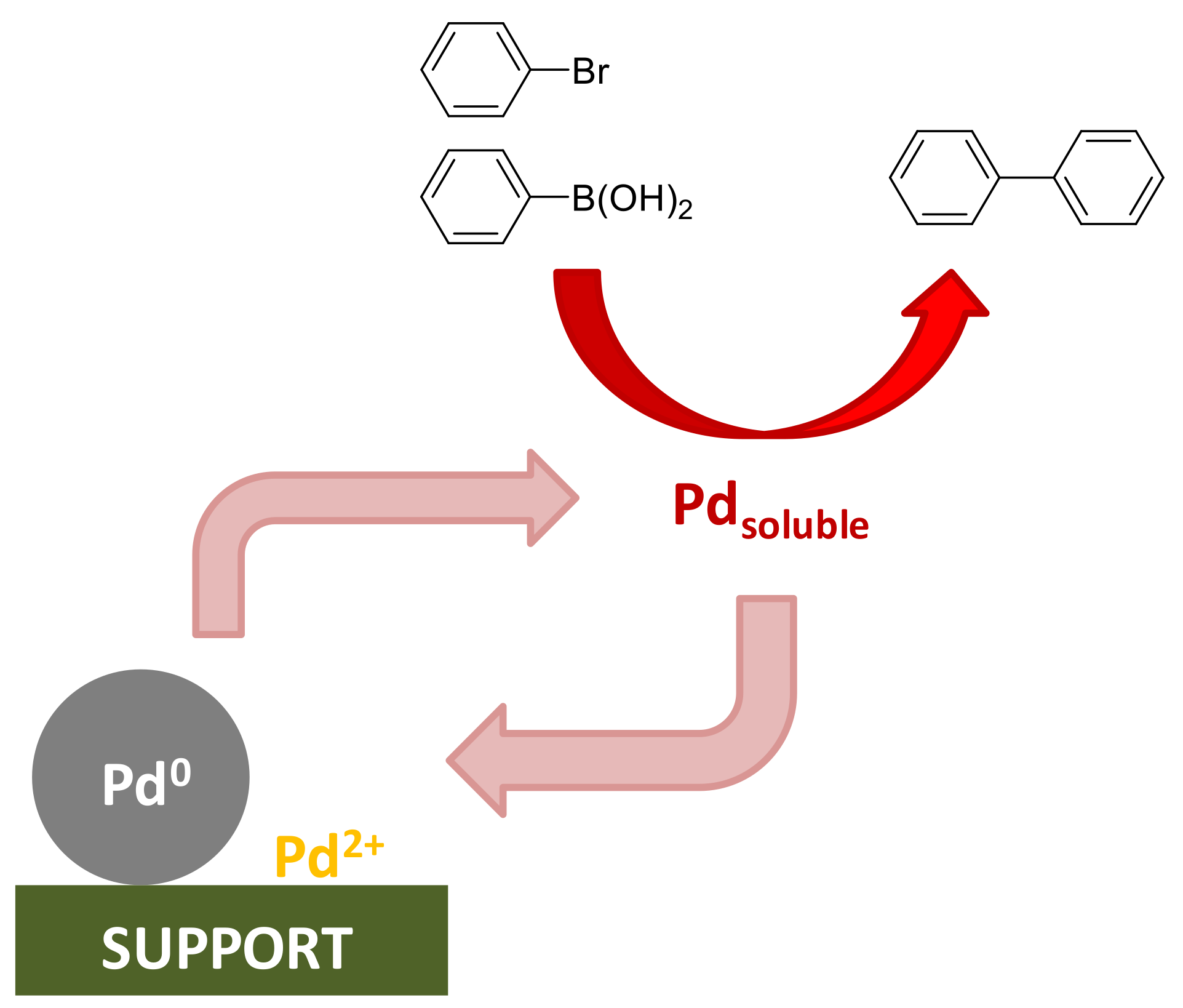
Effect ligand imposes on efficiency of palladium-mediated unprotected... | Download Scientific Diagram

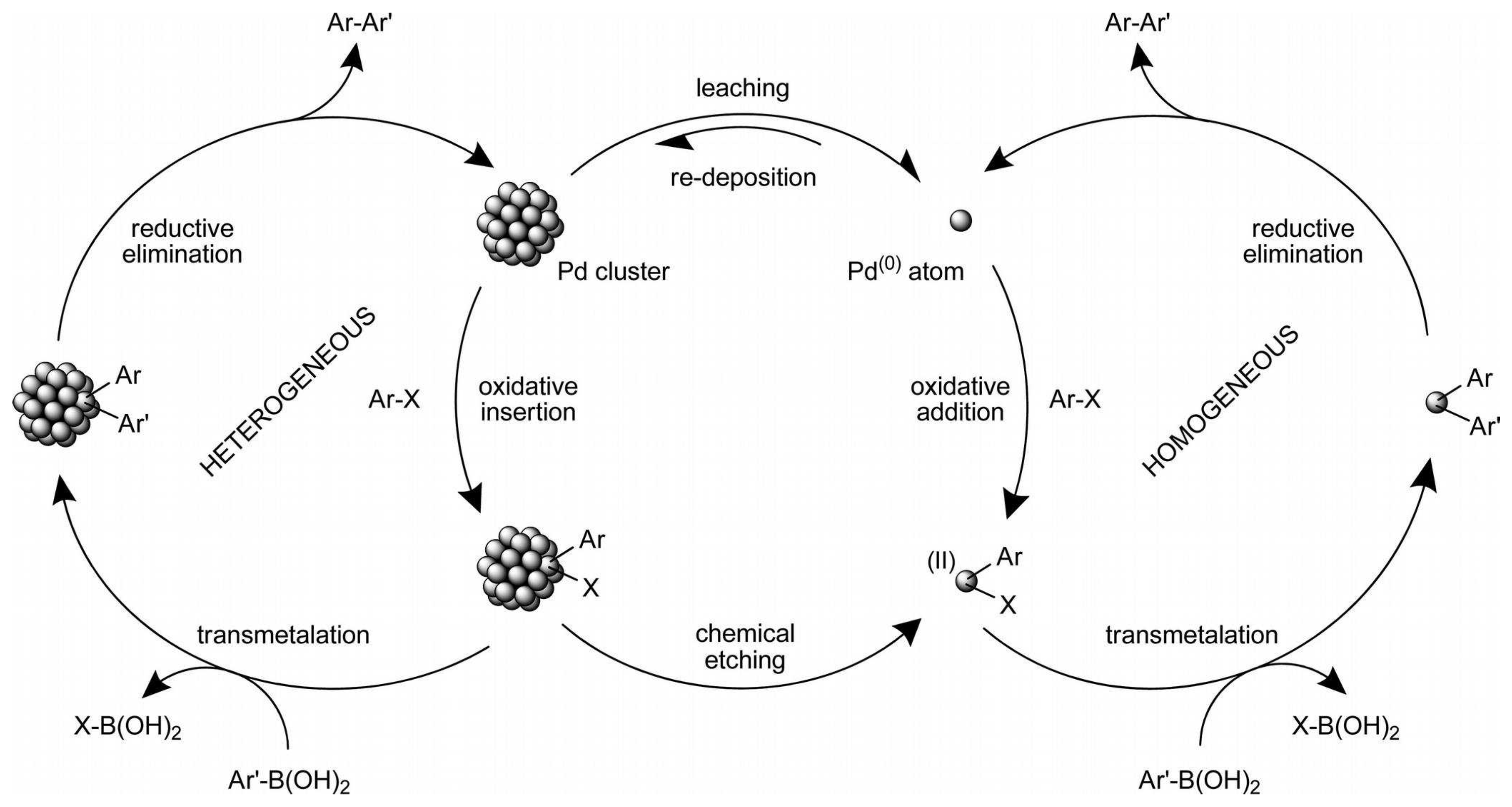
SciELO - Brasil - Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the
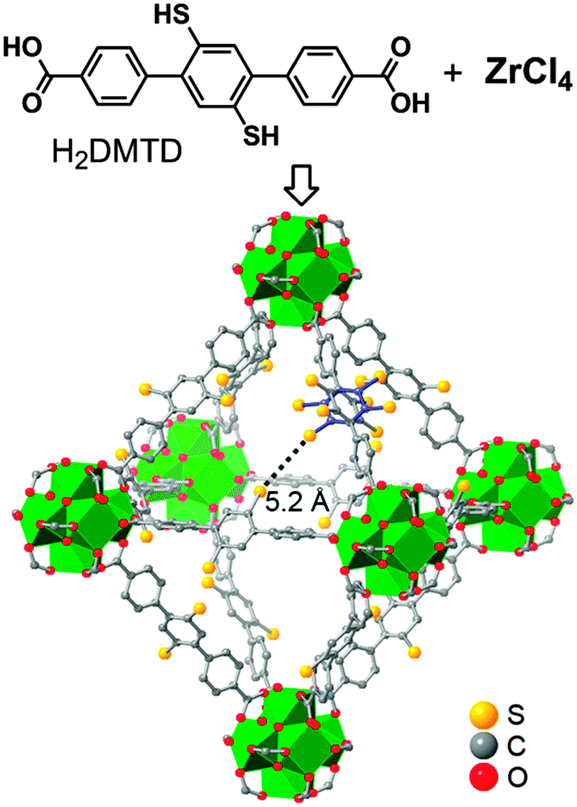
Tackling poison and leach: catalysis by dangling thiol–palladium functions within a porous metal–organic solid - Chemical Communications (RSC Publishing)

Thiol Functionalized Cross-Linked Chitosan Polymer Supporting Palladium for Oxidative Heck Reaction and Reduction of p-Nitrophenol | SpringerLink

Pd/BIPHEPHOS is an Efficient Catalyst for the Pd‐Catalyzed S‐Allylation of Thiols with High n‐Selectivity - Schlatzer - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

A general and efficient method for the palladium-catalyzed cross-coupling of thiols and secondary phosphines
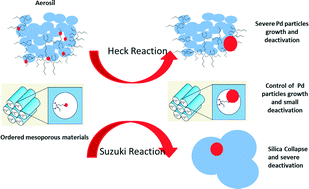
Palladium nanoparticles confined in thiol-functionalized ordered mesoporous silica for more stable Heck and Suzuki catalysts - Catalysis Science & Technology (RSC Publishing)

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect